Повернутися до бібліотеки Ромера
Emerging Mycotoxins – A Threat beyond Regulations?

Mycotoxins are naturally occurring, secondary metabolites produced by various molds. These compounds are toxic to humans and animals. Toxigenic molds contaminate a wide range of crops and produce mycotoxins as a result of the infection of plant tissues in the field. Unfortunately, the formation of these toxins can continue even after harvest and the level of mycotoxins in grains continues to increase during storage. Contaminated crops represent a major health risk to humans and animals. The most prominent mycotoxin-producing field-fungi are represented by fungi of the species Fusarium and Aspergillus. Beyond these, there are over 300 different fungi that are known to produce over 400 different mycotoxins.
In recent years, more and more mycotoxins have been considered as relevant as they contribute to the risk posed to humans and animals. Risk assessment studies have been performed for various important mycotoxin groups including ergot alkaloids (see article on ergot alkaloids), Alternaria toxins (see article on Alternaria toxins) and modified or masked mycotoxins.
Modifying mycotoxins as plant defense
Typically, mycotoxins are explicitly produced by fungi and their parent structure is often modified by the fungus itself which releases a cocktail of structurally related compounds. During infection, these substances are then often further modified by the host plant of the fungus. The living plant might change the chemical structure of toxins and produce so-called masked mycotoxins.
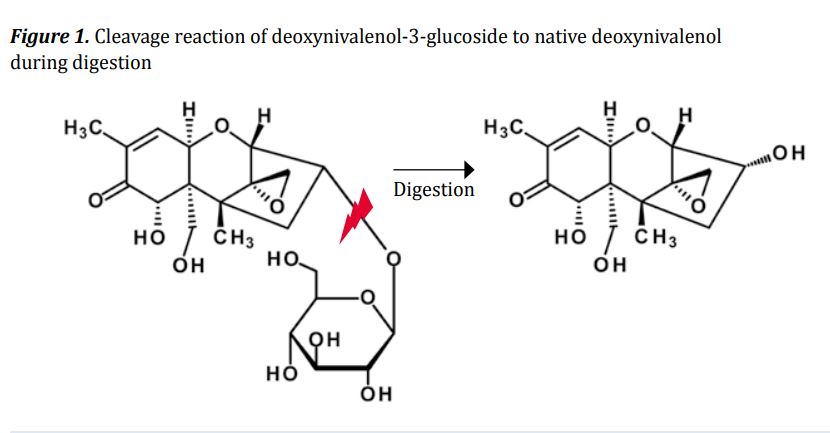
The formation of these masked toxins is a major detoxification strategy of crops, as they are less toxic for the plant. Usually, a glucose molecule or a sulfate is involved in the conjugation and detoxification. Although these masked toxins do not further harm the plant, their toxicity to humans and animals might reemerge when the added masking molecule is cleaved in the gastrointestinal tract of mammals during digestion (Figure 1). In plant breeding, the increasing occurrence and production of some masked mycotoxins might be linked to novel resistant breeds. Deoxynivalenol-3-glucoside, for example, has been reportedly linked to resistance against Fusarium head blight. This means that Fusarium resistant plants have been proven to show higher deoxynivalenol-3-glucoside to deoxynivalenol ratios, but these are accompanied by lower levels of total deoxynivalenol and the modified form due to higher Fusarium resistance.
The term “modified mycotoxin” includes both the modification of a parent toxin molecule by the fungus itself, and the masking of the toxin which only occurs in the plant tissue. Another type of modification takes place in mammals when aflatoxin B1 is consumed through contaminated feed and converted to aflatoxin M1. This aflatoxin M1 migrates into the milk of lactating animals and is excreted with it. In addition, modifications of toxins can also occur during food processing, in particular heating and fermentation, increasing their prevalence. These modified mycotoxins might occur in relevant amounts in food and feed.
The phenomenon of modifying mycotoxins is particularly related to Fusarium toxins (trichothecenes, zearalenone and fumonisins) but modified forms have also been reported for other mycotoxins like aflatoxins, ochratoxin A or patulin.
Altered and masked forms of deoxynivalenol – an example
Deoxynivalenol is the mycotoxin with the most studies conducted on the different versions of frequently observed modifications. The modified forms of deoxynivalenol can be divided into two main groups: alteredand masked forms. There are two main altered forms of deoxynivalenol secreted by the fungus itself: 3-acetyl-deoxynivalenol and 15-acetyl-deoxynivalenol, as found in Fusarium-contaminated cereals. Plants are able to mask the deoxynivalenol to deoxynivalenol-3-glucoside and as recent studies show, this may take on two sulfonated forms: deoxynivalenol-3-sulfateand deoxynivalenol-15-sulfate (Table 1).
How harmful are modified and emerging mycotoxins?
Modified mycotoxins can be either more or less toxic than their parent compounds. For example, they maybe more bio available due to modifications. Toxicological data on modified mycotoxins are scarce, and current results and knowledge on the real risks and effects of these compounds are insufficient. This lack of knowledge makes it difficult to conduct a proper risk assessment. Nevertheless, there have been studies describing their potential threat to food safety. Furthermore, it has to be highlighted that masked mycotoxins can be “unmasked” again in the digestive tract of animals and humans, releasing the parent compound with its toxicological effects again. A similar situation exists with emerging mycotoxins: toxicological data are scarce which makes it difficult to set up regulations and maximum tolerated limits to protect humans and animals from potential health risks.
Do regulations cover all mycotoxin risks?
To ensure food and feed safety, many countries have established regulatory limits for mycotoxins in crops. Currently, in most developed countries, there are regulations on maximum levels or at least guidance levels for mycotoxins in food and feed. These regulations only cover some of the known mycotoxins such as aflatoxins B1, B2, G1, G2 and M1; fumonisins B1, B2 and B3; ochratoxin A, deoxynivalenol, zearalenone, HT-2 toxin andT-2 toxin.
As modified mycotoxins behave differently in their chemical reactions to parent mycotoxins, they can be easily missed in routine analysis. Current detection methods for regulated mycotoxins in food and feed do not include routine screening for these modified mycotoxins as they are not covered by legislation. Such standard methods may show up contamination levels below legislative limits, while contaminations from modified mycotoxins go undetected. This represents a correct result, but from a toxicological point of view the integration of modified toxins (e.g. as a sum parameter) would provide more sound data for risk assessment. Together, all these facts point to the possible hazards posed by modified mycotoxins to human health. Regulations on the maximum levels of modified mycotoxins as well as other emerging mycotoxins are currently under discussion in the European Union.
Analytical methods for mycotoxin quantification
Mycotoxins are commonly analyzed by chromatographic methods like liquid chromatography–mass spectrometry (LC-MS) and immunochemical methods like enzyme-linked immunosorbent assay (ELISA). Immunochemical methods can, depending on the cross-reactivity of the antibody, respond to more than one compound (e.g. native mycotoxins and their modified forms) leading to a single result. In contrast LC based separation methods might underestimate the total toxin levels as those methods resolve each compound as a single parameter and are usually only developed for the parent mycotoxins.
Limits of analytical methods
There are two ways to detect and quantify modified mycotoxins: A “direct” approach that measures the whole modified compound, and an “indirect” approach that measures the parent compound after chemical or enzymatic treatments that lead to the cleavage of modified mycotoxins, mainly by hydrolysis. Among the advantages of the indirect method are that reference materials for modified mycotoxins are not needed for correct quantification, and that all modified forms are included in the final result. The main disadvantages are that the efficiency of the hydrolysis process cannot be verified easily, and that there is no access to the quantities of the different forms of a toxin. Therefore, it is important to develop direct methods to obtain further insight into the occurrence of modified mycotoxins. All chromatographic technologies for parent mycotoxins are also potentially suitable for their modified forms as long as they are soluble and directly availablefor analysis.
One major constraint of the direct determination and quantification of modified mycotoxins is the limited availability of reference materials (pure substancesor calibrants in addition to isotope-labeled internal standards).
Another drawback is that most methods require an adequate cleanup prior to the analysis procedure. Commercially available purification devices are currently designed for native mycotoxins and might not be necessarily suitable for modified forms. Work is currently underway to develop new reference standards as well as innovative cleanup devices to determine modified mycotoxins directly.
Emerging mycotoxins -A threat beyond regulations?
With the current gaps in routine mycotoxin analysis driven by missing regulations for emerging mycotoxins, many of these compounds can go undetected and pose a threat to both human and animal health. The extent of this threat, thought to be considerable, is however hard to estimate since toxicological data is still scarce, despite increasing research efforts in this direction.
Опубліковано:
Mycotoxin